The effects of endogenous melatonin on brain tissue oxidative stress induced by photoperiodic alterations and iron overloading in rats
Melatonin on brain tissue oxidative stress
Abstract
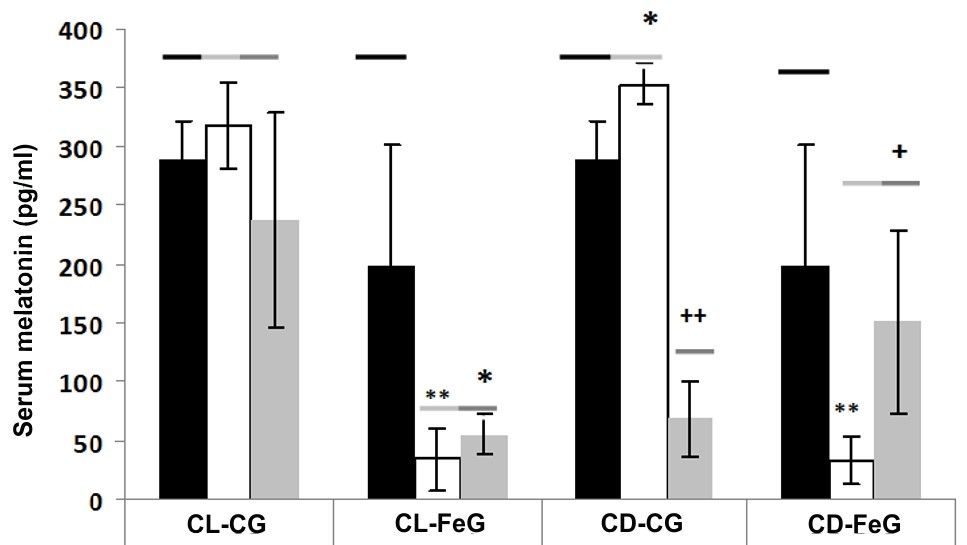
Melatonin is a potent endogenously occurring antioxidant with the pleiotropic activities to neurodegenerative diseases associated with brain oxidative damage. In this study, we examined the prolonged photoperiodic alterations and iron (Fe) overload on melatonin production and brain oxidative stress in rats. The result showed that the 15 days of constant light (CL) exposure did not low the melatonin production but the 15 days of constant darkness (CD) significantly increased serum melatonin level in rats. The Fe treatment in both CL and CD conditions significantly reduced endogenous melatonin levels and increased brain tissue lipid peroxidation. Fe as a toxic transition metal can induce Fenton reaction to generated hydroxyl radical which can damage the neuronal cell membrane and impair the brain antioxidant system. In the current study, we observed the imbalanced antioxidant defense alterations upon Fe treatment in rat brain including the increased levels of alpha-tocopherol (α-T) and total thiols and the reduced melatonin level and catalase (CAT) activity. We speculated that the reduced melatonin level caused by Fe was due to its consumption since melatonin served as a metal chelator and antioxidant. Understanding these aspects enhances knowledge of brain Fe metabolism and its role in neurodegenerative disorders as well as the potential protective effects of melatonin on this metal.
References
2. Wiechmann AF, Sherry DM (2013) Role of melatonin and its receptors in the vertebrate retina. Int. Rev. Cell Mo.l Biol. 300: 211-242. doi: 10.1016/B978-0-12-405210-9.00006-0.
3. Cardinali DP, Pévet P (1998) Basic aspects of melatonin action. Sleep Med. Rev. 2: 175-190.
4. Arendt J (1988) Melatonin. Clin. Endocrinol. 29: 205–229.
5. Reiter RJ (1993) The melatonin rhythm—both clock and calendar. Experientia 49: 654–664. doi: 10.1016/S0079-6123(08)81008-4.
6. Reiter RJ, Tan DX, Fuentes-Broto L (2010) Melatonin: a multitasking molecule. Prog. Brain Res. 181: 127–151.
7. Smith JA, O’Hara J, Schiff AA (1981) Altered diurnal serum melatonin rhythm in blind men. Lancet 2: 933–938.
8. Lewy AJ, Wehr TA, Goodwin FK, Newsome DA, Markey SP (1980) Light suppresses melatonin secretion in humans. Science 210: 1267–1269.
9. González MMC (2018) Dim light at night and constant darkness: Two frequently used lighting conditions that jeopardize the health and well-being of laboratory rodents. Front. Neurol. 9: 609. https://doi.org/10.3389/fneur.2018.00609.
10. Cervino CO, Cogo Pagella J, Hernando MP (2023) Effects of long-term exposure to light or darkness and return to normal light-dark cycle on serum melatonin levels in rats. Melatonin Res. 6(2): 215-223. doi: 10.32794/mr112500150.
11. Tordjman, S, Chokron, S, Delorme R, Charrier A, Bellissant E, Jaafari N, Fougerou C (2017) Melatonin: Pharmacology, functions and therapeutic benefits. Cur. Neuropharmacol. 15: 434-443.
12. Arendt J, Skene DJ (2005) Melatonin as a chronobiotic. Sleep Med. Rev. 9: 25–39. doi: 10.1016/j.smrv.2004.05.002.
13. Pandi-Perumal SR, Srinivasan V, Maestroni GJM, Cardinali DP, Poeggeler B, Hardeland R (2006) Melatonin—Nature’s most versatile biological signal? FEBS J. 273: 2813–2838. DOI: 10.1111/j.1742-4658.2006.05322.x.
14. Tan DX, Hardeland R, Manchester LC, Paredes SD, Korkmaz A, Sainz RM, Mayo JC, Fuentes-Broto L, Reiter RJ (2010). The changing biological roles of melatonin during evolution: from an antioxidant to signals of darkness, sexual selection and fitness. Biol. Rev. Camb. Philos. Soc. 85: 607–623. doi: 10.1111/j.1469-185X.2009.00118.x.
15. Garcia JJ, Lopez-Pingarron L, Almeida-Souza P, Tres A, Escudero P, Garcia-Gil FA, Tan DX, Reiter RJ, Ramirez JM, Bernal-Perez M (2014) Protective effects of melatonin in reducing oxidative stress and in preserving the fluidity of biological membranes: a review. J. Pineal Res. 56: 225–237. doi: 10.1111/jpi.12128.
16. Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J. Physiol. 552 (Pt 2): 335-344. doi: 10.1113/jphysiol.2003.049478.
17. González PM, Malanga G, Puntarulo S (2015) Cellular oxidant/antioxidant network: update on the environmental effects over marine organisms. Open Marine Biol. J. 9 (1): 1-13. http://dx.doi.org/10.2174/1874450801509010001.
18. Jones DP (2006) Redefining oxidative stress. Antioxid. Redox. Signal. 8: 1865–1879. doi: 10.1089/ars.2006.8.1865.
19. Halliwell B, Gutteridge JMC (2007). Free radicals in biology and medicine. Oxford University Press, New York. 4th Edition, pp. 936.
20. Padurariu M, Ciobica A, Lefter R, Serban IL, Stefanescu C (2013) The oxidative stress hypothesis in Alzheimer’s disease. Psychiatr. Danub. 25 (4): 401–409.
21. Imlay JA (2003) Pathways of oxidative damage. Annu. Rev. Microbiol. 57 (1): 395-418. doi: 10.1146/annurev.micro.57.030502.090938.
22. Reiter RJ, Tan DX, Manchester LC, Qi W (2001) Biochemical reactivity of melatonin with reactive oxygen and nitrogen species: a review of the evidence. Cell Biochem. Biophys. 34: 237–256. doi: 10.1385/CBB:34:2:237.
23. Reiter RJ, Tan DX, Mayo JC, Sainz RM, Leon J, Czarnocki Z (2003) Melatonin as an antioxidant: biochemical mechanisms and pathophysiological implications in humans. Acta Biochim. Pol. 50 (4): 1129-1146.
24. Galano A, Tan DX, Reiter RJ (2013) On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 54 (3): 245–257.
25. Reiter R, Tang L, Garcia JJ, Munoz-Hoyos A (1997) Pharmacological actions of melatonin in oxygen radical pathophysiology. Life Sci. 60: 2255–2271.
26. León J, Escames G, Rodríguez MI, López LC, Tapias V, Entrena A, Camacho E, Carrión MD, Gallo MA, Espinosa A, Tan DX, Reiter RJ, Acuña-Castroviejo D (2006) Inhibition of neuronal nitric oxide synthase activity by N1-acetyl-5-methoxykynuramine, a brain metabolite of melatonin. J. Neurochem. 98: 2023–2033. https://doi.org/10.1111/j.1471-4159.2006.04029.x.
27. Tan DX, Manchester LC, Terron MP, Flores LJ, Reiter RJ (2007) One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 42: 28–42.
28. Zhou JN, Liu RY, Kamphorst W, Hofman MA, Swaab DF (2003) Early neuropathological Alzheimer’s changes in aged individuals are accompanied by decreased cerebrospinal fluid melatonin levels. J. Pineal Res. 35: 125–130.
29. Wu YH, Swaab DF (2005) The human pineal gland and melatonin in aging and Alzheimer’s disease. J. Pineal Res. 38: 145–152.
30. Lin L, Huang QX, Yang SS, Chu J, Wang JZ, Tian Q (2013) Melatonin in Alzheimer’s disease. Int. J. Mol. Sci. 14 (7): 14575–14593. doi: 10.3390/ijms140714575.
31. Antolin I, Mayo JC, Sainz RM, del Brio ML, Herrera F, Martin V, Rodriguez C (2002) Protective effect of melatonin in a chronic experimental model of Parkinson’s disease. Brain Res. 943: 163–173.
32. Paredes SD, Reiter RJ (2010) Melatonin: Helping cells cope with oxidative disaster. Cell Memb. Free Radic. Res. 2 (3): 99-111.
33. Emerit J, Edeas M, Bricaire F (2004) Neurodegenerative diseases and oxidative stress. Biomed. Pharmacother. 58: 39–46. doi: 10.1016/j.biopha.2003.11.004.
34. Symons MCR, Gutteridge JMC (Eds). (1998). Free Radicals and Iron: Chemistry, Biology and Medicine. Oxford University Press, New York. pp. 258.
35. Gutteridge JM, Halliwell B (2000) Free radicals and antioxidants in the year 2000: A historical look to the future. Ann. N. Y. Acad. Sci. 899 (1): 136-147. doi: 10.1111/j.1749-6632.2000.tb06182.x.
36. Dexter DT, Wells FR, Lees AJ, Agid F, Agid Y, Jenner P, Marsden CD (1989) Increased nigral iron content and alterations in other metal ions occurring in brain in Parkinson's disease. J. Neurochem. 52 (6): 1830–1836.
37. Markesbery WR (1997) Oxidative stress hypothesis in Alzheimer's disease. Free Radic. Biol. Med. 23 (1): 134–147. doi: 10.1016/s0891-5849(96)00629-6.
38. Willmore LJ, Sypert GW, Munson JB (1978) Recurrent seizures induced by cortical iron injection: a model of posttraumatic epilepsy. Ann. Neurol. 4 (4): 329–336. doi: 10.1002/ana.410040408.
39. Hidalgo C, Carrasco MA, Muñoz P, Nuñez MT (2007) A Role for Reactive Oxygen/Nitrogen Species and Iron on Neuronal Synaptic Plasticity. Antioxid. Redox. Signal. 9 (2): 245-255. doi: 10.1089/ars.2007.9.245.
40. Sofic E, Paulus W, Jellinger K, Riederer P, Youdim MBH (1991) Selective increase of iron in substantia nigra zona compacta of parkinsonian brains. J. Neurochem. 56 (3): 978–982.
41. Thompson CM, Markesbery WR, Ehmann WD, Mao YX, Vance DE (1988) Regional brain trace element studies in Alzheimer's disease. Neurotoxicology 9 (1): 1–7.
42. Bello-Caraballo N, Cogo Pagella J, Iodice O, Cervino CO (2018) Efectos de la combinación de melatonina con clorhidrato de ketamina sobre regímenes estándar de anestesia en ratas. Rev. Argent. Anestesiol. 76: 75-84. doi: 10.24875/RAA.21000011.
43. Cogo Pagella J, Hernando MP, Cervino CO (2023) Effect of iron on rat serum melatonin levels under different light/dark cycle patterns. Melatonin Res. 6 (2): 148-160. doi: 10.32794/mr112500146.
44. Mantovani M, Kaster MP, Pertile R, Calixto JB, Rodrigues AL, Santos AR (2006) Mechanisms involved in the antinociception caused by melatonin in mice. J. Pineal Res. 41 (4): 382-389. doi: 10.1111/j.1600-079X.2006.00380.x.
45. Ayer RE, Sugawara T, Chen W, Tong W, Zhang JH (2008) Melatonin decreases mortality following severe subarachnoid hemorrhage. J. Pineal Res. 44 (2): 197-204. doi: 10.1111/j.1600-079X.2007.00508.x.
46. Pechlivanova D, Dzambazova E, Kolev G, Petkova Z, Tchekalarova J (2016) Effects of melatonin on stress-induced and diurnal variations of nociception in wistar and spontaneously hypertensive rats. C. R. Acad. Bulg. Sci. 69 (9): 1223-1230.
47. Fukuhara C, Aguzzi J, Bullock N, Tosini G (2005) Effect of long-term exposure to constant dim light on the circadian system of rats. Neurosignals 14: 117-125.
48. Piloni NE, Reiteri M, Hernando MP, Cervino CO, Puntarulo S (2017). Differential effect of acute iron overload on oxidative status and antioxidant content in regions of rat brain. Toxicol. Pathol. 45 (8): 1067-1076.
49. Boveris AD, Puntarulo S (1998) Free-radical scavenging actions of natural antioxidants. Nutrition Res. 18 (10): 1545–1557.
50. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193 (1): 265–275.
51. Malanga G, Puntarulo S (1995) Oxidative stress and antioxidant content in Chlorella vulgaris after exposure to ultraviolet-B radiation. Physiologia Plantarum 94 (4): 672–679. https://doi.org/10.1111/j.1399-3054.1995.tb00983.x.
52. Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal. Biochem. 25 (1): 192–205.
53. Aebi H (1984) Catalase in vitro. Methods Enzymol. 105: 121-126.
54. Piloni NE, Fermandez V, Videla LA, Puntarulo S (2013) Acute iron overload and oxidative stress in brain. Toxicology 314 (1): 174-182.
55. Lockman JA, Geldenhuys WJ, Bohn KA, Desilva SF, Allen DD, Van der Schyf CJ (2012) Differential effect of nimodipine in attenuating iron-induced toxicity in brain- and blood-brain barrier-associated cell types. Neurochem. Res. 37 (1): 134-142. doi: 10.1007/s11064-011-0591-2.
56. Triggs W J, Willmore LJ (1984) In vivo lipid peroxidation in rat brain following intracortical Fe2+ injection. J. Neurochem. 42 (4): 976–980.
57. Sloot WN, van der Sluijs-Gelling AJ, Gramsbergen JBP (1994) Selective lesions by manganese and extensive damage by iron after injection into rat striatum or hippocampus. J. Neurochem. 62 (1): 205–216.
58. Cini M, Fariello RY, Bianchettei A, Moretti A (1994) Studies on lipid peroxidation in the rat brain. Neurochem. Res. 19 (3): 283-288.
59. Häubner N, Sylvander P, Vuori K, Snoeijs P (2014) Abiotic stress modifies the synthesis of alpha-tocopherol and beta-carotene in phytoplankton species. J. Phycol. 50: 753–759. https://doi.org/10.1111/jpy.12198
60. Liu Z, Zhou T, Ziegler AC, Dimitrion P, Zuo L (2017) Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxid. Med. Cell Longev. 2017: 2525967. doi: 10.1155/2017/2525967.
61. Hou YJ, Zhao YY, Xiong B, Cui XS, Kim NH, Xu YX, Sun SC (2013) Mycotoxin-containing diet causes oxidative stress in the mouse. PLoS One 8 (4), e60374.
62. Kotler M, Rodriques C, Sainz RM, Antolin I, MenendezPelaes A (1998) Melatonin increases gene expression for antioxidant enzymes in rat brain cortex. J. Pineal Res. 24: 83–89.
63. Barlow-Walden LR, Reiter RJ, Abe M, Pablos M, Menendez-Pelaes A, Chen LD, Poeggeler B (1995) Melatonin stimulates brain glutathione peroxidase activity. Neurochem. Int. 26 (5): 497–502. doi: 10.1016/0197-0186(94)00154-m.
64. Tan DX, Chen LD, Poeggeler B, Manchester LC, Reiter RJ (1993) Melatonin: a potent endogenous hydroxyl radical scavenger. Endocr. J. 1: 57–60.
65. Pieri C, Marra M, Moroni F, Recchioni R, Marcheselli F (1994) Melatonin: a peroxyl radical scavenger more effective than vitamin E. Life Sci. 55 (4): 271–276.
66. Limson J, Nyokong T, Daya S (1998) The interaction of melatonin and its precursors with aluminum, cadmium, copper, iron, lead, and zinc: an adsorptive voltammetric study. J. Pineal Res. 24 (1): 15–21.
67. Acuña-Castroviejo D, Lopez LC, Escames G, Lopez A, Garcia JA, Reiter RJ (2011) Melatonin-mitochondria interplay in health and disease. Curr. Top Med. Chem. 11 (2): 221–240. doi: 10.2174/156802611794863517.
68. Reiter R, Paredes S, Korkmaz A, Manchester L, Tan D (2008) Melatonin in relation to the "strong" and "weak" versions of the free radical theory of aging. Advances in Medical Sciences 53 (2): 119-129. doi: 10.2478/v10039-008-0032-x.
69. Maharaj DS, Maharaj H, Daya S, Glass BD (2006) Melatonin and 6-hydroxymelatonin protect against iron-induced neurotoxicity. J. Neurochem. 96 (1): 78–81. doi: 10.1111/j.1471-4159.2005.03532.x.
70. Zang LY, Cosma G, Gardner H, Vallynathan V (1998) Scavenging of reactive oxygen species by melatonin. Biochim. Biophys. Acta 1425 (3): 467-477. doi: 10.1016/s0304-4165(98)00099-3.
71. Tan DX, Manchester LC, Reiter RJ, Qi WB, Karbownik M, Calvo J R (2000) Significance of melatonin in antioxidative defense system: reactions and products. Neurosignals 9 (3-4): 137-159.
72. Montilla Lopez P, Tunez I, Munoz de Agueda MC, Cabrera E, Montitla MC, Plascencia J, de la Torre EJ (2000) Protective effect of melatonin against oxidative stress induced by ligature of extra-hepatic bilim3, duct in rats: Comparison with the effect of s-adenosyl-L-methionine. J. Pineal Res. 28: 143-149.
73. Viña J (1990) Glutathione: Metabolism and Physiological Functions. CRC Press, Boca Raton. pp. 392.
74. Giblin FJ, David LL, Wilmarth PA, Leverenz VR, Simpanya M F (2013) Shotgun proteomic analysis of sthiolation sites of guinea pig lens nuclear crystallins following oxidative stress in vivo. Mol. Vis. 19: 267–280.
75. Ji Y, Akerboom TP, Sies H, Thomas JA (1999) S-nitrosylation and s-glutathiolation of protein sulfhydryls by s-nitroso glutathione. Arch. Biochem. Biophys. 362 (1): 67–78. doi: 10.1006/abbi.1998.1013.
76. Baba SP, Wetzelberger K, Hoetker JD, Bhatnagar A (2009) Posttranslational glutathiolation of aldose reductase (akr1b1): A possible mechanism of protein recovery from s-nitrosylation. Chem. Biol. Interact. 178 (1–3): 250–258. doi: 10.1016/j.cbi.2008.11.007.
77. Hill BG, Bhatnagar A (2012) Protein s-glutathiolation: Redox-sensitive regulation of protein function. J. Mol. Cell. Cardiol. 52 (3): 559–567. doi: 10.1016/j.yjmcc.2011.07.009.
78. Kaiserova K, Tang XL, Srivastava S, Bhatnagar A (2008). Role of nitric oxide in regulating aldose reductase activation in the ischemic heart. J. Biol. Chem. 283 (14): 9101–9112. doi: 10.1074/jbc.M709671200.
79. Conway ME, Coles SJ, Islam MM, Hutson SM (2008) Regulatory control of human cytosolic branched-chain aminotransferase by oxidation and s-glutathionylation and its interactions with redox-sensitive neuronal proteins. Biochemistry 47 (19): 5465–5479. doi: 10.1021/bi800303h.
80. Regazzoni L, Panusa A, Yeum KJ, Carini M, Aldini G (2009) Hemoglobin glutathionylation can occur through cysteine sulfenic acid intermediate: Electrospray ionization ltq-orbitrap hybrid mass spectrometry studies. J. Chromatogr B Analyt. Technol. Biomed. Life Sci. 877 (28): 3456–3461. doi: 10.1016/j.jchromb.2009.05.020.
81. West MB, Hill BG, Xuan YT, Bhatnagar A (2006). Protein glutathiolation by nitric oxide: An intracellular mechanism regulating redox protein modification. FASEB J. 20 (10): 1715–1717.
82. Tappel AL (1973) Vitamin E. Nutrition Today 8: 4-12.
83. McCluskey S, Hall M, Stanton C, Devery R. (1999) Alpha-tocopherol inhibits oxidative stress induced by cholestanetriol and 25-hydroxycholesterol in porcine ovarian granulosa cells. Mol. Cell. Biochem. 194 (1-2): 217-225. doi: 10.1023/a:1006967219894.
84. Glatz JFC, Luiken JJFP, Van Nieuwenhoven FA, Van der Vusse G J (1997). Molecular mechanism of cellular uptake and intracellular translocation of fatty acids. Prostaglandins, Leukot. Essent.Fatty Acids 57 (1): 3-9. https://doi.org/10.1016/S0952-3278(97)90485-3.
85. Qian J, Morley S, Wilson K, Nava P, Atkinson J, Manor D (2005) Intracellular trafficking of vitamin E in hepatocytes: The role of tocopherol transfer protein. J. Lipid Res. 46 (10): 2072-2082. doi: 10.1194/jlr.M500143-JLR200.

This work is licensed under a Creative Commons Attribution 4.0 International License.
For all articles published in Melatonin Res., copyright is retained by the authors. Articles are licensed under an open access Creative Commons CC BY 4.0 license, meaning that anyone may download and read the paper for free. In addition, the article may be reused and quoted provided that the original published version is cited. These conditions allow for maximum use and exposure of the work, while ensuring that the authors receive proper credit.
In exceptional circumstances articles may be licensed differently. If you have specific condition (such as one linked to funding) that does not allow this license, please mention this to the editorial office of the journal at submission. Exceptions will be granted at the discretion of the publisher.


